Message on the topic my favorite substance is water. Abstract: Properties of water
INTRODUCTION
Water is the most common substance on our planet. Oceans, seas and rivers, glaciers and atmospheric water - - that's far from full list"reservoirs" of water on Earth. Even in the bowels of our planet there is water, and what can we say about living organisms living on its surface! There is not a single living cell that does not contain water. The human body, for example, consists of more than 70% water.
Life on Earth is a combination of numerous complex processes, the main place among which is the circulation of heat, moisture and substances. main role This is played by water - the progenitor of life on Earth.
But is it accidental that our life is inseparable from water, and what are the reasons for this?
Unlike ordinary people, who are accustomed to considering water as something so mundane and familiar that it is not worth much thought, and even more so surprise, scientists consider this liquid the most mysterious and amazing. For example, many properties of water are anomalous, that is, they differ significantly from the corresponding properties of compounds of a similar structure. Oddly enough, but it was the anomalous properties of water that gave this liquid the opportunity to become the most important on Earth.
WATER IN NATURE
In a free state, the Earth contains a colossal amount of water - about one and a half billion cubic kilometers. Almost the same amount of water is in the physical and chemical bound state in crystalline and sedimentary rocks.
Most natural waters are solutions, the content of dissolved substances in which ranges from 0.01% (in fresh water) to 3.5% (in sea water).
Fresh water accounts for only about 3% of the total water supply on the planet (approximately 35 million km3). A person can directly use only 0.006% of fresh water for his needs - this is that part of it that is contained in the channels of all rivers and lakes. The rest of fresh water is difficult to access - 70% are ice sheets of the polar regions or mountain glaciers, 30% are underground aquifers.
It is no exaggeration to say that our planet is saturated with water. Precisely, thanks to this, the development of those forms of life that we see around us became possible on Earth.
PROPERTIES OF WATER,
CONTRIBUTE TO THE APPEARANCE OF LIFE ON EARTH
Comparing the properties of water with the properties of analogue compounds, we come to the conclusion that many characteristics of water have anomalous values. As will be said below, it is this anomalous property that will play the most important role for the origin and existence of life on Earth.
Boiling temperature
Consider the boiling points of compounds of the H2El series, where El is an element of the main subgroup of group VI.
Compound H 2 0 H 2 S H 2 Se H 2 Te
t°C b.p. +100 -60 -41 -2
As can be seen, the boiling point of water differs sharply from the boiling point of compounds of analogous elements and has an abnormally high value. It has been established that a similar anomaly is observed for all compounds of the H 2 El type, where El is a strongly electronegative non-metal (O, N, etc.).
If in the series H 2 Te-H 2 Se-H 2 S the boiling point decreases uniformly, then from H 2 S to H 2 0 it increases abruptly. The same is observed for the series HI -HBr-HCl-HF and H 3 Sb-H 3 As-H 3 P-H 3 N. It was assumed, and subsequently proved, that there are specific bonds between H 2 0 molecules, the breaking of which consumes energy heating. The same bonds make it difficult to detach the HF and H 3 N molecules. This type of bond is called a hydrogen bond, let's look at its mechanism.
The elements H and O have a large difference in electronegativity values (EO(H) = 2.1; EO(O) = 3.5), so the chemical N-O bond heavily ruffled. The electron density shifts towards oxygen, as a result of which the hydrogen atom acquires an effective positive charge, and oxygen atom acquires an effective negative charge. A hydrogen bond is an image of the electrostatic attraction between the positively charged hydrogen atom of one molecule and the negatively charged oxygen atom of another molecule:
The ability of water to form hydrogen bonds is of great biochemical importance.
Density
All substances tend to increase in density with decreasing temperature. However, water in this case behaves somewhat unusually.
The minimum temperature at which water can be without freezing is 0 "C. It would be logical to assume that the highest density of water also corresponds to this temperature. However, it has been experimentally proven that the density of liquid water is maximum at 4 ° C.
This fact is of great importance. Imagine that water obeys the laws that are characteristic of all other liquids. Then the change in its density would occur, as in other liquids. In the world around us, this would lead to disaster: with the approach of winter and widespread cooling, the upper layers of liquid in reservoirs would cool down and sink to the bottom. The warmer layers of liquid that had risen to take their place would also have cooled to 0 °C and descended. This would continue until all the water had cooled to 0°C. Further, the water, starting from the upper layers, would begin to freeze. Being denser, the ice would sink to the bottom, freezing would continue until all the water of natural reservoirs froze to the bottom. It is clear that in such conditions the flora and fauna of natural water bodies could not exist.
Another anomaly in the density of water is that the density of ice is lower than the density of water, i.e., water does not compress when freezing, like all other liquids, but, on the contrary, expands.
From the point of view of the laws of physics, this is absurd, because a more ordered state of molecules (ice) cannot occupy a larger volume than a less ordered one ( liquid water) provided that the number of molecules in both states is the same.
As already mentioned, in liquid water, H 2 0 molecules are interconnected by hydrogen bonds. The formation of ice crystals is accompanied by the formation of new hydrogen bonds, as a result of which water molecules form layers. The connection between the layers is also carried out due to hydrogen bonds. The resulting structure (the so-called ice structure) is one of the least dense - the voids that exist between the molecules in an ice crystal exceed the size of a water molecule. Therefore, the density of water is of greater importance than the density of ice.
Surface tension
As a rule, the surface tension of a liquid is understood as a force acting on a unit length of the interface contour and tending to reduce this surface to a minimum. The value of surface tension for water has an abnormally high value - 7.3 .10 -2 N / m at 20 0 C (of all liquids, only mercury has a higher value - 51 10 -2 N / m).
The high value of the surface tension of water is manifested in the fact that it tends to reduce its surface to a minimum. It can be said that under the action of this force, the molecules of the outer layer of water adhere, forming a kind of film on the surface. It is so strong and resilient that individual objects have the ability to stay on the surface of the water without sinking into it, even if their density more density water.
The presence of a film enables many insects to move on the surface of the water and even sit on it, as on a hard surface.
Inner side The surface of the water is also actively used by living beings. Many of us have seen mosquito larvae hanging on it or small snails crawling in search of prey.
High surface tension also causes such an unusually important phenomenon in nature as capillarity (liquid rises through very thin tubes - capillaries). Thanks to this, plant nutrition is carried out.
To describe the behavior of water in capillaries, quite complex physical laws have been derived. Water layers located near a solid surface are structurally ordered. The thickness of such a layer can reach tens and hundreds of molecules. Now scientists are inclined to consider the structurally ordered state of water in capillaries as a separate state - capillary.
Capillary water is widely distributed in nature in the form of so-called pore water. It covers the surfaces of pores and cracks in rocks and minerals with a thin but dense film. earth's crust. The density of this film is also due to the fact that its constituent water molecules are associated with particles that form solid, intermolecular forces. The structural ordering of pore water is the reason that its crystallization (freezing) temperature is noticeably lower than the temperature of free water. In addition, the properties of rocks with which pore water comes into contact depend significantly on the state of aggregation it is in.
Water on our planet is in three states - liquid, solid (ice, snow) and gaseous (steam). Currently, water occupies 3/4.
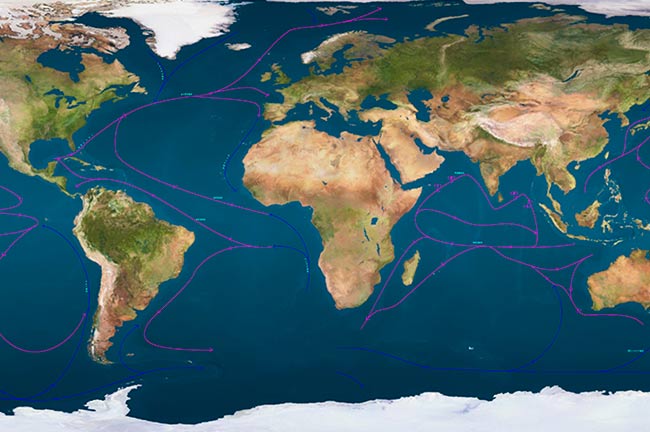
Water forms the water shell of our planet - the hydrosphere.
The hydrosphere (from the Greek words "hydro" - water, "sphere" - a ball) includes three main components: the oceans, land waters and water in the atmosphere. All parts of the hydrosphere are interconnected by the process of the water cycle in nature already known to you.
- Explain how water from the continents enters the oceans.
- How does water get into the atmosphere?
- How does water get back to land?
The oceans account for over 96% of all water on our planet.
Continents and islands divide the World Ocean into separate oceans: Pacific, Atlantic, Indian,.
In recent years, the maps highlight the Southern Ocean - the body of water surrounding Antarctica. The largest in area - Pacific Ocean, the smallest is the Arctic.
The parts of the oceans that protrude into the land and differ in the properties of their waters are called seas. There are a lot of them. largest seas planets - Philippine, Arabian, Coral.
Water in natural conditions contains various substances dissolved in it. In 1 liter of ocean water, on average, contains 35 g of salt (most of all table salt), which gives it a salty taste, makes it unsuitable for drinking and use in industry and agriculture.
Rivers, lakes, swamps, glaciers and underground waters are land waters. Most of the land's waters are fresh, but salty ones are also found among lakes and groundwater.
You know what a huge role rivers, lakes, swamps play in nature and people's lives. But here's what is surprising: in the total amount of water on Earth, their share is very small - only 0.02%.
Much more water is enclosed in glaciers - about 2%. Do not confuse them with the ice that forms when water freezes. occur where more falls than has time to melt. Gradually, the snow accumulates, compacts and turns into ice. Glaciers cover about 1/10 of the land. They are located primarily on the mainland of Antarctica and the island of Greenland, which are covered with huge ice shells. Blocks of ice that break off along their shores form floating mountains - icebergs.
Some of them reach enormous sizes. Large areas are occupied by glaciers in the mountains, especially in such high places as the Himalayas, the Pamirs, and the Tien Shan.

Glaciers can be called pantries of fresh water. So far, it has hardly been used, but scientists have long been developing projects to transport icebergs to arid regions in order to provide local residents with drinking water.
They also make up about 2% of all water on Earth. They are located in the upper part of the earth's crust.
These waters can be salty and fresh, cold, warm and hot. Often they are saturated with substances useful for human health and are medicinal (mineral waters).
In many places, for example, along the banks of rivers, in ravines, groundwater comes to the surface, forming springs (they are also called springs and springs).
Groundwater reserves are replenished due to atmospheric precipitation, which seeps through some of the rocks that make up earth's surface. Thus, groundwater is involved in nature.
Water in the atmosphere
Contains water vapor, water droplets and ice crystals. Together they make up fractions of a percent of the total amount of water on Earth. But without them, the water cycle on our planet would be impossible.
- What is the hydrosphere? List its constituent parts.
- What oceans form the World Ocean of our planet?
- What makes up land water?
- How are glaciers formed and where are they located?
- What is the role of groundwater?
- What is water in the atmosphere?
- What is the difference between river, lake and ?
- What is the danger of an iceberg?
- Are there salt water bodies on our planet other than seas and oceans?
The water shell of the Earth is called the hydrosphere. It consists of the oceans, land waters and water in the atmosphere. All parts of the hydrosphere are interconnected by the process of the water cycle in nature. The oceans account for over 96% of the world's water. It is divided into separate oceans. The parts of the oceans that jut out into the land are called seas. Land waters include rivers, lakes, swamps, glaciers, groundwater. The atmosphere contains water vapor, water droplets and ice crystals.
I would be grateful if you share this article on social networks:
Site search.
Custom Search
Everything interesting about water
Added: 2012-12-11
Everything interesting about water

Water is the simplest and most common substance on the planet. But at the same time, water is fraught with many mysteries.
The human need for water is second only to oxygen. We need water to live! As you know, a person can live up to 6 weeks without food, but only one week without water.
The chemical compound H2O does not exist in its pure form in nature. Water is a universal solvent, it dissolves in itself everything that it meets on the way. For example, enjoying the taste of well water, we pour into ourselves a “compote”, the composition of which, perhaps, no one could reproduce.
The most expensive water in the world is sold in Los Angeles (USA). Its price is $ 90 per liter. According to sellers, the water has an ideal pH balance for humans and an exquisite taste. The liquid is sold in special bottles decorated with Swarovski crystals. Water has gained great popularity among wealthy youth, who line up for it in the most expensive nightclubs in California. Among those wishing to purchase exclusive bottles, Hollywood stars were also noticed.
A person drinks an average of 35 tons of water in his life.

Water can reduce the risk of a heart attack if a person drinks more than five glasses of water a day. However, only pure drinking water has such properties.
Water should be drunk more if you are lovers of alcoholic beverages, as well as tea or coffee. Since when they are consumed, the kidneys work at full capacity, creating a risk of dehydration.
In Yugoslavia there is a lake Tsirknitskoe. In it, the water disappears in summer and winter, and in spring and autumn it returns with the fish.
Water is the only substance that occurs in nature in three forms: solid (ice), liquid and gas. By the way, only 3% of the water on earth is fresh, but at the same time most of fresh water is contained in a frozen state in glaciers.
The writer Griboyedov in one of his works said: "It is difficult to forget the taste of the melt water of high-mountain streams." It is believed that it is thanks to the melt water that the highlanders live so long.
Some carbonated drinks contain harmful chemicals that cause dehydration rather than quench your thirst.

One of the most watery foods is watermelon. Watermelon is 93% water.
Approximately 70 percent of the Earth is covered with water. But only 1 percent of this water is drinkable!
In nature, there are about 1330 types of water. They differ in origin (for example: rain, soil, from fresh snow, etc.), they also differ in the amount of substances dissolved in it.
American nutritionists recommend getting rid of excess weight with the help of a “water” diet, which is becoming increasingly popular in the West. It was found that with a lack of fluid, the body begins to send signals to the brain similar to those of hunger. As a result, people start eating when they should be drinking! By drinking 2-2.5 liters of water every day, a person can suppress hunger and reduce the amount of food consumed. But it is water that you need to drink, since even tea or coffee contains active substances harmful to the diet.
There is a lake of ink in Algiers. You can write on paper with the water of this lake and the ink never runs out.
There is a lake in Antarctica, the water in which is 11 times saltier than sea water and can only freeze at t -50 degrees C
A 2% reduction in body water levels can result in a 20% decrease in mental and physical performance.
In total, there are about three million cubic kilometers of fresh water on Earth. Every day, 1 trillion tons of water evaporate from the earth's surface.
If all the water in the world contained in various reservoirs is poured into the open ocean, then its level will rise by 3 cm. By the way, in the last 100 years, the ocean level around the world has been rising by more than a millimeter per year.
According to doctors, water helps to increase blood pressure. And also, as it turned out, The best way get rid of edema - drink plenty of water.
The human body is 60-70% water, and a child's body is 80%. A five-month-old embryo consists of 94% water.
According to experts, it is enough to drink two glasses of clean water to overcome depression and fatigue.
Water delivers nutrients (vitamins, mineral salts) to the cells of the body and carries away waste products. In addition, water is involved in the process of thermoregulation and respiration.

According to UNESCO, the most pure water in the world - in Finland. The study, conducted in 122 countries around the world, took into account the quality and quantity of fresh water.
Worldwide, 1 billion 100 million people do not have access to water. So we are lucky people!
There is combustible water in Azerbaijan! From the match, the water lights up with a blue flame due to methane.
The most dangerous water in the world is in Sicily in one of the lakes. From the bottom of the lake, 2 sources of concentrated acid are beating, poisoning the water.
A cubic centimeter of sea water contains 1.5 grams of protein and many other nutrients. Scientists have calculated that Atlantic Ocean"in nutritional value" is estimated at 20,000 harvests per year on the entire land.
Recent experiments have shown that water energizes food, after which food particles transfer this energy to the body during digestion. It turns out that food without water has absolutely no energy value for the body.
About 840 liters of water per day flow out of your needle-wide faucet.
Cheers!
Most of our planet - 79% - is occupied by water, and even if you delve into the thickness of the earth's crust, you can find water in cracks and pores. In addition, all minerals and living organisms known on Earth contain water.
The importance of water in nature is great. Modern Scientific research waters make it possible to consider it as a unique substance. It participates in all physical-geographical, biological, geochemical and geophysical processes occurring on the Earth, is the driving force of many global processes on the planet.
Water has caused such a phenomenon on Earth as The water cycle - a closed, continuous process of water movement, covering all the most important shells of the Earth. driving force The water cycle is powered by solar energy, which causes the evaporation of water (from the oceans 6.6 times more than from the land). Water entering the atmosphere is carried by air currents in a horizontal direction, condenses and falls to the Earth in the form of precipitation under the influence of gravity. One part of them enters the lakes and the ocean through the rivers, and the other goes to moisten the soil and replenish groundwater, which take part in the nutrition of rivers, lakes and seas.
525.1 thousand km 3 of water are involved in the annual cycle. On average, 1030 mm of precipitation falls on our planet per year and about the same amount evaporates (525,000 km 3 in volume units).
The equality between the amount of water entering the surface of the Earth with precipitation and the amount of water evaporating from the surface of the oceans and land for the same period of time is called water balance our planet (Table 19).
Table 19 Water balance Lands (according to M.I. Lvovich, 1986)
For the evaporation of water, a certain amount of heat is required, which is released when the water vapor condenses. Consequently, the water balance is closely related to the heat balance, while the moisture circulation evenly distributes heat between its spheres, as well as the regions of the Earth, which has great importance for all geographical envelope.
The importance of water and economic activity. It is impossible to list all areas of human activity in which water is used: domestic and industrial water supply, irrigation, electricity generation, and many others.
The largest biochemist and mineralogist academician V. I. Vernadsky noted that water stands apart in the history of our planet. Only she can stay on Earth in three states of aggregation and move from one to another (Fig. 158).
Water, which is in all states of aggregation, forms the water shell of our planet - hydrosphere.
Since water is contained in the lithosphere, the atmosphere and in various living organisms, it is very difficult to determine the boundaries of the water shell. In addition, there are two interpretations of the concept of "hydrosphere". In a narrow sense, the hydrosphere is a discontinuous water shell Earth, consisting of the World Ocean and inland water bodies. The second interpretation - broad - defines it as a continuous shell of the Earth, consisting of open reservoirs, water vapor in the atmosphere and groundwater.

Rice. 158. Aggregate states of water
Water vapor in the atmosphere is called the diffuse hydrosphere, and groundwater is called the buried hydrosphere.
As for the hydrosphere in the narrow sense, most often the surface is taken as its upper boundary. the globe, and the lower boundary is drawn along the level of groundwater, which is located in the loose sedimentary strata of the earth's crust.
When considering the hydrosphere in a broad sense, its upper boundary is located in the stratosphere and is very indefinite, that is, it lies above the geographical envelope that does not extend beyond the troposphere.
Scientists say that the volume of the hydrosphere is approximately 1.5 billion km 3 of water. The vast majority of the area and volume of water falls on the oceans. It contains 94% (according to other sources, 96%) of the volume of all water contained in the hydrosphere. About 4% is the buried hydrosphere (Table 20).
Analyzing the volumetric composition of the hydrosphere, one cannot limit oneself to one quantitative side. When evaluating the component parts of the hydrosphere, its activity in the water cycle should be taken into account. To this end, the famous Soviet hydrologist, Doctor of Geographical Sciences M.I. Lvovich introduced the concept water exchange activity, which is expressed as the number of years required for a full renewal of the volume.
It is known that in all the rivers on our planet the simultaneous volume of water is small and amounts to 1.2 thousand km 3. At the same time, channel waters are completely renewed on average every 11 days. Almost the same activity of water exchange is characteristic of the diffuse hydrosphere. But underground waters, the waters of the polar glaciers of the ocean, require millennia to be completely renewed. The water exchange activity of the entire hydrosphere is 2800 years (Table 21). The lowest activity of water exchange in polar glaciers is 8000 years. Since in this case the slow water exchange is accompanied by the transition of water to a solid state, the masses polar ice constitute preserved hydrosphere.
Table 20. Distribution of water masses in the hydrosphere
|
Parts of the hydrosphere |
Share in world reserves, % |
||
|
from the total water supply |
from fresh water reserves |
||
|
World Ocean |
|||
|
The groundwater |
|||
|
Glaciers and permanent snow cover |
|||
|
including in Antarctica |
|||
|
Groundwater in the permafrost zone |
|||
|
including fresh lakes |
|||
|
Water in the atmosphere |
|||
|
Total fresh water resources |
|||
|
Total water supply |
|||
Table 21
* Taking into account the underground runoff into the ocean, bypassing the rivers: 4200 lay.
Table 21
The hydrosphere has come a long way of evolution, repeatedly changing in mass, ratio separate parts, the movement of an ox, the ratio of dissolved gases, suspensions and other components, the changes of which are recorded in the geological record, which is far from being fully deciphered.
When did the hydrosphere appear on our planet? It turns out that it existed already at the very beginning of the geological history of the Earth.
As we already know, about 4.65 billion years ago, the Earth arose. The oldest rocks found are 3.8 billion years old. They kept their imprints unicellular organisms who lived in water bodies. This allows us to judge that the primary hydrosphere appeared no later than 4 billion years ago, but it was only 5-10% of its modern volume. According to one of the most common hypotheses today, water appeared during the formation of the Earth by melting and degassing of the mantle matter(from lat. negative particles de and French gas- gas) - the removal of dissolved gases from the mantle. Most likely, the impact (catastrophic) degassing of the mantle substance, caused by the fall of large meteorite bodies to the Earth, initially played a large role.
Initially, the increase in the volume of the surface hydrosphere proceeded very slowly, since a significant part of the water was spent on other processes, including the addition of water to minerals (hydration, from the Greek. hydro- water). The volume of the hydrosphere began to grow intensively after the rate of release of rocks waters exceeded the rate of their accumulation. At the same time, there was an entry into the hydrosphere juvenile waters(from lat. juvenilis- young) - godzmnyx waters formed from oxygen and hydrogen released from magma.
Water is still released from magma, falling on the surface of our planet during volcanic eruptions, during the formation of the oceanic-type earth's crust in the zones of stretching of lithospheric plates, and this will continue to happen for many millions of years. The volume of the hydrosphere now continues to grow at a rate of about 1 km 3 of water per year. In this regard, it is assumed that the volume of the water mass of the World Ocean will increase by 6-7% over the next billion years.
Based on this, until quite recently, people were sure that water supplies would last forever. But in fact, due to the rapid pace of consumption, the amount of water is drastically reduced, and its quality has also drastically decreased. Therefore, one of the most important problems today is the organization of the rational use of water and their protection.
Water in human life
Water - at first glance, the simplest chemical compound two hydrogen atoms and one oxygen atom - is, without any exaggeration, the basis of life on Earth. It is no coincidence that scientists are looking for life forms on other planets. solar system so much effort goes into detecting traces of water.
In our Everyday life we are in contact with water all the time. At the same time, paraphrasing a song from an old movie, we can say that we “drink water” and “pour water”. We will talk about these two aspects of human use of water.
Water "food"
Household water
Water "food"
Water itself has no nutritional value, but it is essential integral part all living things. Plants contain up to 90% water, while the body of an adult consists of about 60 - 65% of it. Looking into the details, it can be noted that the bones contain 22% water, the brain 75%, while the blood consists of as much as 92%.
The primary role of water in the life of all living beings, including humans, is due to the fact that it is a universal solvent. huge amount chemical substances. Those. in fact, it is the environment in which all life processes take place.
Here is just a small and far from complete list of the "duties" of water in our body.
Regulates body temperature.
Humidifies the air.
Provides delivery of nutrients and oxygen to all cells of the body.
Protects and buffers vital organs.
Helps convert food into energy.
Helps nutrients to be absorbed by the organs.
Removes toxins and waste products of life processes.
A certain and constant water content is here necessary condition the existence of a living organism. When the amount of water consumed and its salt composition change, the processes of digestion and assimilation of food, hematopoiesis, etc. are disrupted. environment and maintenance of body temperature.
A person is extremely acutely aware of the change in the water content in his body and can live without it for only a few days. With a loss of water in an amount of less than 2% of body weight (1-1.5 l), a feeling of thirst appears, with a loss of 6-8%, a fainting state occurs, with 10% - hallucinations, swallowing disorders. Loss of 10-20% of water is life-threatening. Animals die when they lose 20-25% of water.
Excessive water consumption leads to an overload of the cardiovascular system, causes debilitating sweating, accompanied by a loss of salts, and weakens the body.
Depending on the intensity of work, external conditions (including climate), cultural traditions, a person consumes in total (along with food) from 2 to 4 liters of water per day and the same amount of water is excreted from the body (for more details, see “Drinking regimen and balance of water in the body" and the article "To drink or not to drink - that is the question" from the journal "Health" in our "Digest"). The average daily consumption is about 2-2.5 liters. It is from these figures that the World Health Organization (WHO) proceeds when developing recommendations for water quality (See "Water Quality Parameters").
The mineral composition of the water is of no small importance. Suitable for constant drinking and cooking fresh water with total mineralization up to 0.5 - 1 g/l. Although, of course, limited quantities it is possible (and sometimes even useful, for example, for medicinal purposes) to use mineral water with a high salt content (for information on which water is “suitable” for which diseases, see the article “Water for each disease” in our Digest). The human body quickly adapts to changes in the salt composition of drinking water. However, the process of getting used to takes some time. Therefore, with a sharp (and even more frequent) change in the characteristics of water, disturbances in the activity of the gastrointestinal tract, popularly known as "travelers' disease", are possible.
In general, the question of what useful substances and in what quantities should be contained in water in products mass media given a lot of attention. This problem is really very important, but, unfortunately, there is too much speculation and profanity around it.
Even very reputable publications allow themselves to somewhat irresponsibly publish information such as: “a person receives up to 25% of useful minerals from water” and other, to put it mildly, information that does not quite correspond to reality. A classic of the genre “I heard a ringing, but I don’t know where it is” - the article “Capital Water ...” by Mrs. Ekaterina Bychkova in AiF-Moscow No. 37 "99.
Our point of view on this issue can be found in the section “Water and useful minerals”.
We also recommend a series of articles from the magazine "Health": "To drink or not to drink - that is the question", "Each disease has its own water", "Five facts about water that you did not know", as well as materials "Both heals and cripples ” and “Stone Waterfall”, also presented in our Digest.
Household water
It is well known that the use of water for household purposes in Russia is far from rational (we tactfully keep silent about industry for lack of reliable data). There are two main reasons:
abundance water resources.
Their cheapness.
In its issue of August 31, 1999, devoted to the problems of water, the magazine "Itogi" provided visual data characterizing these two parameters and their relationship.
It can be seen that the cheaper water is in a particular country, the more generously it is poured. It is also not surprising that in Russia, where before recent years there was no practice of installing water metering devices for each apartment, and there is no reliable statistics on water consumption in everyday life.
Therefore, we will use published English data from the mid-80s. Of course, in the UK, the daily water consumption per capita was already at that time 140 l / day, and in our country it is still around 400 l / day, but the data collected by the meticulous British is so interesting that we should study it and take note . In any case, the market economy dictates its own laws, it is likely that water will soon rise in price and the thriftiness of the aforementioned Englishmen will no longer seem unreasonable to us.
So. According to English data /15/:
The main article of water consumption in everyday life is the toilet. The "gentle contralto of the water-tank instrument" is responsible for 35% of water consumption per capita per day (50 liters). Next comes personal hygiene (bathing and showering, washing, etc.) - 32% of consumption (45 l), laundry - 12% (17 l), washing dishes - 10% (14 l), drinking and cooking - 3% (4 l), other expenses (pets, watering flowers, etc.) - 8% (11 l).
It is clear that these figures are averaged and reduced to one day (for example, a person takes a bath and does not wash every day). However, they also provide food for thought and comparisons with our reality.
It is unlikely that we eat much more than the same British and, accordingly, we spend on cooking also somewhere between 4 - 4.5 liters per capita per day. Forgive us for such a conclusion, but it directly follows from the previous one that we should not use the toilet more often (or are there other opinions?). Considering that we have a single European standard for drain tanks, this gives the same 50 liters.
By the way, the meticulous British calculated that a family of two adults and three children on average uses the toilet 25-40 times a day. If there is a habit of flushing leftover food and other waste down the toilet, then the number of “descents” even in a family of 4 people can reach 60. Here, by the way, one should look for the origins of the now fashionable in Europe (especially in Scandinavia) ecological initiative “Give a brick in the toilet bowl!”. In addition to jokes, they put a brick in the tank, thus reducing the volume of water in it by almost 2 liters. Multiply by the number of flushes per day and get a "net" savings. And if we are talking about such an interesting area of human life as a toilet, then the future is generally behind vacuum units (similar to those installed in airplanes), which consume only 1 (one) liter of water per session.
But back to our sheep. We also venture to suggest that in terms of the level of automation of washing, we still reached the level of England 15 years ago, and for this purpose the average per capita consumption is 17 liters.
Where, then, as our first president used to say, "the dog rummaged"? Why do we use twice as much water?
To do this, let's see what is left of the water consumption items: personal hygiene, washing dishes, and so on. Here, perhaps, lies the solution. It's not that we bathed more and washed the dishes more thoroughly. The difference is rather that we do not have the habit of turning off the faucet when, for example, we brush our teeth, and we also wash dishes in running water. It would seem - a trifle, but keep in mind that 10-15 liters of water flow out through an open tap per minute. And the second powerful "reserve" is the position "Other". The fact is that “they” in this section practically do not have such an article as leaks. It's just that life makes them quickly repair the current plumbing - not only water flows, money flows. We can with with good reason argue that in our conditions the lion's share of leaks occurs precisely in homes, so to speak, already "after the meter". And that's why.
The British pay great attention to leaks, but for the reasons stated, their main leaks occur in the municipal water supply network. In Moscow, according to experts, 15-16% of water is also lost between the water intake station and the apartment (see the article “Moscow Water Drinkers”, magazine “Itogi”, 08/31/99). And now, attention, the most important thing. It's not that bad, but just a great result! In England, losses average 25%, and their experts, recognizing the inevitability of leaks, believe that a realistically achievable result to which one should strive for leaks is 15%. Which, as they say, was required to be proved. Honor and praise to Mosvodokanal. We suspect, however, that the average situation in the country is rather closer to the English one. However, even if this is true, it still once again shows where we are losing. We, unfortunately, are used to blaming everything on the water supply, but it turns out that "there is nothing to blame on the mirror ...". It's time to understand that after the pipes have entered the building (be it a residential building, an office center or an industrial facility), the responsibility already lies with the owners and users.
So, you see, in the near future we will also need a brick in the toilet bowl and other “bourgeois” tricks. As the same British say: "Warned out is already armed."
